Living things contain a large percentage of water, so they demonstrate diamagnetic behavior. Water, like most molecules, contains all paired electrons. We can calculate the number of unpaired electrons based on the increase in weight. When we compare the weight of a sample to the weight measured in a magnetic field ( Figure 8.27), paramagnetic samples that are attracted to the magnet will appear heavier because of the force exerted by the magnetic field. Magnetic susceptibility measures the force experienced by a substance in a magnetic field. And yet, the Lewis structure of O 2 indicates that all electrons are paired. Such attraction to a magnetic field is called paramagnetism, and it arises in molecules that have unpaired electrons. Thus, when we pour liquid oxygen past a strong magnet, it collects between the poles of the magnet and defies gravity, as in Figure 8.1. By itself, O 2 is not magnetic, but it is attracted to magnetic fields. However, this picture is at odds with the magnetic behavior of oxygen. There is an O=O double bond, and each oxygen atom has eight electrons around it. This electronic structure adheres to all the rules governing Lewis theory. We would write the following Lewis structure for O 2: However, one of the most important molecules we know, the oxygen molecule O 2, presents a problem with respect to its Lewis structure. Relate these electron configurations to the molecules’ stabilities and magnetic propertiesįor almost every covalent molecule that exists, we can now draw the Lewis structure, predict the electron-pair geometry, predict the molecular geometry, and come close to predicting bond angles.Write molecular electron configurations for first- and second-row diatomic molecules.Calculate bond orders based on molecular electron configurations.Describe traits of bonding and antibonding molecular orbitals.Outline the basic quantum-mechanical approach to deriving molecular orbitals from atomic orbitals. 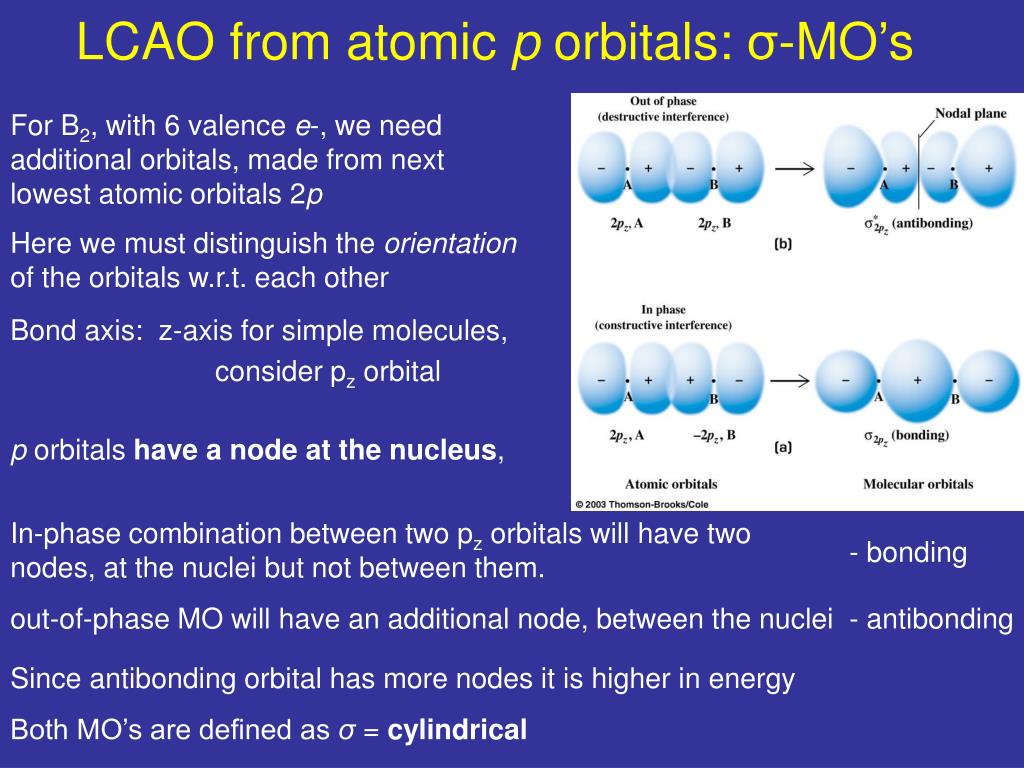
The two is orbitals are in phase and have the same signs.By the end of this section, you will be able to: The formation of two molecular orbitals from two is orbitals is show below. The formation of bonding molecular orbital can be considered as the result of constructive interference of the atomic orbitals and the formation of anti-bonding molecular orbital can be the result of the destructive interference of the atomic orbitals.Ħ. The wave function for molecular orbitals, \(\psi_A\) and \(\psi_B\) can be obtained by the LCAO as shown below: One is bonding molecular orbitai (ψ bonding) and the other is anti-bonding molecular orbital (ψ anti-bonding).Ĥ. 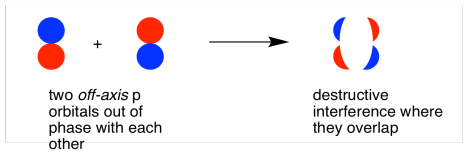
Consider two atomic orbitals represented by the wave functions \(\psi_A\) and \(\psi_B\) with comparable energy that combines to form two molecular orbitals.ģ. Atomic orbitals are represented by wave functions ψ. Since solving Schrodinger wave equation is too complex, a most common method linear combination of atomic orbitals (LCAO) is used to obtain wave function for molecular orbitals.Ģ. The wave functions for the molecular orbitals can be obtained by solving Schrodinger wave equation for the molecule. Linear combination of atomic orbitals (LCAO):ġ. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
