 In the second column, the source of β-lac was from the cytoplasmic cell extract of transiently transfected HeLa cells (with the concentration determined by mCherry intensity), whereas in the third column purified recombinant β-lac produced in Escherichia coli was added to the drop (at a defined concentration). In comparison, the second and third columns show data obtained from analyzing the enzymatic activity within a drop of eukaryotic cell extract using the same microscope settings as for in-cell measurements (see also supplemental Fig. ), we determined apparent k cat/ Km values for CCF2 catalysis from individual cells transfected with TEM1 ( Fig. In the absence of β-lac, no substrate degradation or product accumulation was observed ( Fig. Only cells where the enzyme decreased by less than 20% during the time of recording were chosen for analysis (implying that the cell is intact after injection). 1 A) within the cytoplasm was selected in the individual cells for analysis ( Fig. In the recorded images a region of interest (ROI yellow circles in Fig. ∼2 s after injection the substrate was homogenously distributed throughout the cytoplasm, and within 1–3 min it was fully converted to product. The high CCF2 intensity seen outside the cell at time 0 is due to constant release of material by the injector to the media and has no bearing toward the CCF2 injected into the cell, because CCF2-FA (Free Acid) is cell-impermeant. We have previously shown that transiently expressed CFP-β-lac in HeLa cells is fully active, because it binds its inhibitor protein-BLIP to completion (Īt time 0, CCF2 was microinjected into the cytoplasm and product formation was followed with a confocal microscope simultaneously at the mCherry channel (excitation, 559 nm emission, 575–675 nm) and the CCF2 product channel (excitation, 405 nm emission, 425–475 nm Fig. The fused protein mCherry-β-lac was uniformly expressed in HeLa cells by transient transfection of the gene ( Fig. 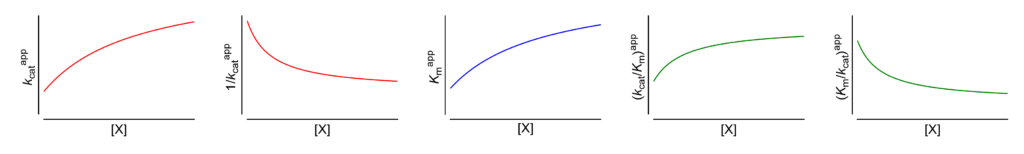
The amount of β-lac was then calculated from the fluorescence intensity of mCherry (excitation, 559 nm emission, 575–675 nm) according to a calibration curve ( supplemental Fig. To measure the β-lac concentration, the enzyme was fused to mCherry without affecting its catalytic efficiency (as determined in vitro using purified proteins). Calibration was done under the microscope using the same setup as for in-cell measurements but performed in a drop where the substrate concentration is controlled. A calibration curve relating product fluorescence with its concentration is shown in supplemental Fig. CCF2 is a fluorogenic substrate of β-lac, which emission peak shifts from 520 to 447 nm upon cleavage ( supplemental Fig. These findings indicate that in vitro data cannot be simply extrapolated to the crowded in vivo environment.Įxperimental determination of catalytic constants requires enzyme and substrate/product concentrations to be known. This suggests substrate-limited reaction rates to be common. The octanol/water partition of the substrate is 4.5, which is in the range of Food and Drug Administration–approved drugs. Simulations showed that for attenuated diffusion the substrate flux becomes rate-limiting, explaining why reaction rates in vivo can be independent on enzyme concentrations. To rationalize these findings, we measured enzyme and substrate diffusion rates in the cell and found the latter to be slower than expected. Surprisingly, the results show that inside the cell the apparent catalytic efficiency decreases, and Km increases with increasing enzyme concentration. We found the apparent in vivo catalytic efficiency, k cat/ Km, to be lower than in vitro, with significant cell-to-cell variability. Here, we determined in real time the catalytic activity of TEM1-β-lactamase inside living cells and compared the values to those obtained in vitro. Glycobiology and Extracellular Matricesįor over a century, enzymatic activity has been studied in vitro, assuming similar activity in the crowded cellular milieu. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
